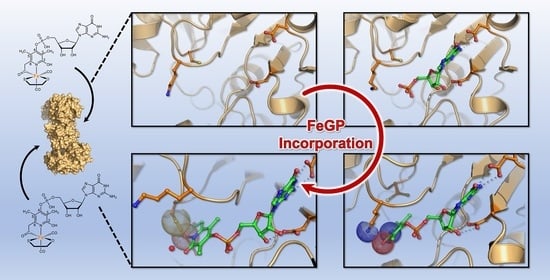
Objective: Model the incorporation of the FeGP cofactor into the hydrogenase and its catalytic capabilities. Context: Fe-hydrogenase is the third type of hydrogenase which requires a cofactor, the FeGP cofactor. The kinetics of incorporation, activation, and catalysis aren't yet fully understood. This work was done as part of the following study: Crystal Structures of [Fe]-Hydrogenase from Methanolacinia paynteri Suggest a Path of the FeGP-Cofactor Incorporation Process
Significance: Understanding cofactor binding and enzyme activation kinetics is an ongoing problem in biochemistry, with various approaches being developed. Mathematical modeling and computational simulation are among these approaches, applied here for the first time to the anaerobic Fe-hydrogenase.
Goal: Determine if observed kinetics can be explained through a mathematical model which further expands the reconstitution hypothesis.
- Gangfeng Huang
- Francisco J. Arriaza G.
- Tristan Wagner
- Seigo Shima
The project consists of several Python files for each condition studied. The kinetic approximation and ODEs derivation can be found in /plots/ODEs_S7 and ODEs_S9, as stated in the supplementary figures of the publication. One master script with the corresponding ODEs is found under /models/hcr.py
Experimental kinetic data performed at the Microbial Protein Structure group led by Dr. Seigo Shima at the Max Planck Institute for Terrestrial Microbiology.
Crystal Structures of [Fe]-Hydrogenase from Methanolacinia paynteri Suggest a Path of the FeGP-Cofactor Incorporation Process.
Gangfeng Huang, Francisco Javier Arriaza-Gallardo, Tristan Wagner and Seigo Shima